Certificate of Achievement
For laboratory assistants and associate practitioners, supporting your training and helping you develop the knowledge and skills to become a valuable member of the laboratory team
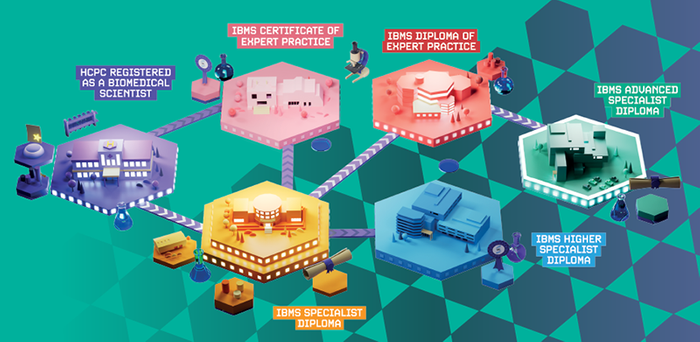
From entry-level to senior roles, IBMS qualifications
equip you with the knowledge and skills to advance
your career in biomedical science
For laboratory assistants and associate practitioners, supporting your training and helping you develop the knowledge and skills to become a valuable member of the laboratory team
Demonstrate the HCPC Standards of Proficiency and qualify for HCPC registration as a biomedical scientist by completing an IBMS Registration Training Portfolio and obtaining an IBMS Certificate of Competence
If you are working at the level of a HCPC-registered biomedical scientist but are not yet registered, you can achieve HCPC registration by completing the IBMS Certificate of Competence by Equivalence
An experiential pathway to HCPC registration as a clinical scientist for professionals working in UK healthcare science

For early-career biomedical scientists to showcase their training, practical skills, specialist knowledge, and competency in a chosen discipline
A professional qualification for early-career biomedical scientists working in research or reference laboratories
For biomedical scientists at Member or Fellow level with at least two years of post-registration experience to specialise and demonstrate their knowledge and skill
Professional qualifications for biomedical scientists with at least five years of post-registration experience working in highly specialised areas
For biomedical scientists aiming to progress to a more advanced role and to demonstrate high levels of knowledge, skill, and competence in their chosen discipline
For biomedical scientists working in research, reference laboratories, or areas outside the main biomedical science disciplines
For advanced practitioners to showcase their expert knowledge and skills at the highest level and achieve formal recognition for their exceptional expertise

To undertake IBMS laboratory-based portfolio qualifications, laboratories must have current IBMS training approval

Contribute to the profession while developing your own expertise and become an IBMS Verifier or Examiner

The digital home for biomedical scientist pre-registration training and aligned to the updated HCPC Standards of Proficiency